Benzoin Can Be Reduced With Reagents Such as Nabh4. What Stereochemical Products Are Possible
Sodium Borohydride (NaBHiv) For Reduction Of Aldehydes And Ketones (And Demercuration)
In a breathy plug for the Reagent Guide, each Friday I profile a dissimilar reagent that is unremarkably encountered in Org 1/ Org ii. Version 1.ii just got released, with a host of corrections and a new folio index.
Having just talked about the oxidation ladder, information technology makes sense to start going into reagents for oxidation and reduction reactions.
Sodium borohydride (NaBHiv)
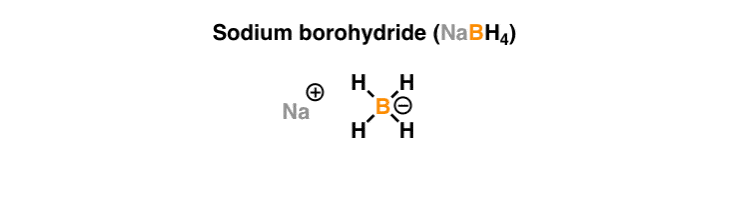
What it'south used for: Sodium borohydride is a good reducing agent. Although not as powerful every bit lithium aluminum hydride (LiAlH4), information technology is very effective for the reduction of aldehydes and ketones to alcohols. Past itself, it will more often than not not reduce esters, carboxylic acids, or amides (although it volition reduce acyl chlorides to alcohols). It is too used in the 2d step of the oxymercuration reaction to replace mercury (Hg) with H.
Similar to: lithium aluminum hydride (LiAlH4) although less reactive.
Reduction Of Aldehydes And Ketones With NaBH4
For our purposes, sodium borohydride is really useful for ane thing: it will reduce aldehydes and ketones. In this sense it traverses one rung on the oxidation ladder. Hither are some examples of information technology in action.
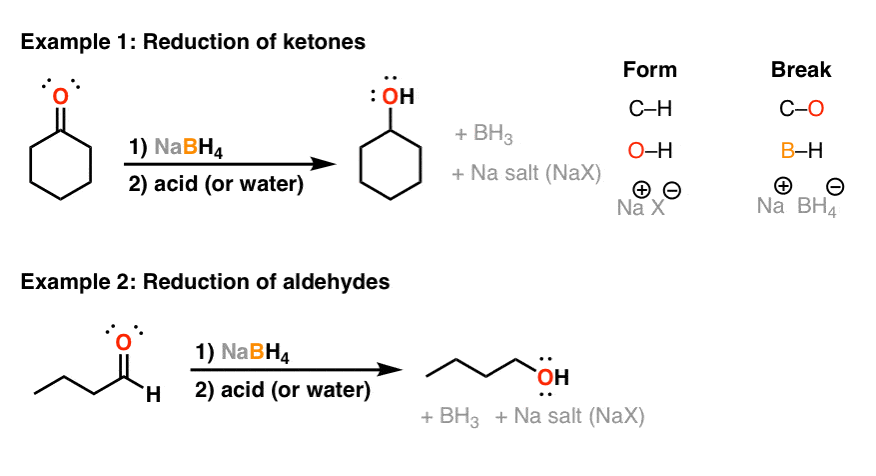
Notice the pattern: nosotros are breaking a C-O bail and replacing information technology with a C-H bond. This is what helps u.s. classify the reaction as a reduction.
Note that nosotros also course an O-H bond. This is where textbooks and other sources are sometimes not equally clear equally they should exist: in gild to make the alcohol, the oxygen needs to option upward a proton (H+) from either water or acid that is added after the reaction is complete (note: this is often referred to as the workup).
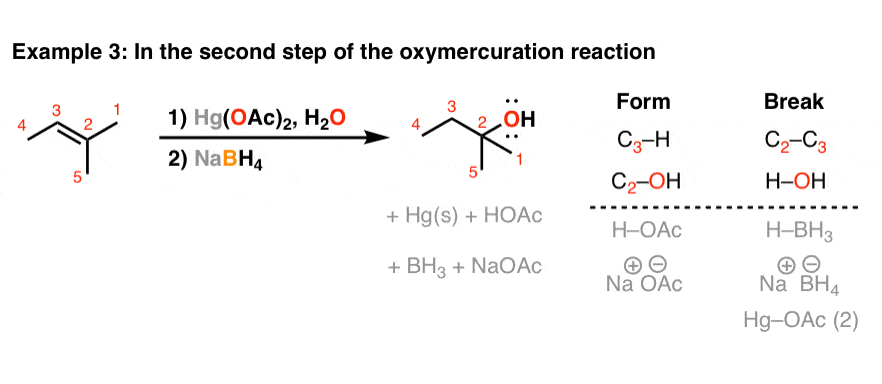
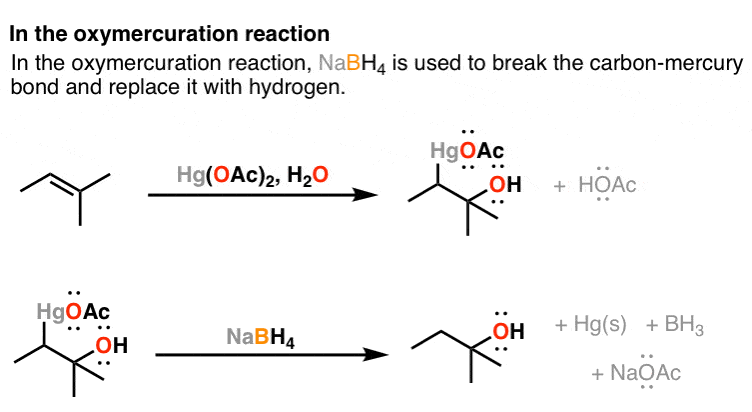
Sodium Borohydride For "Demercuration" (The Second Step Of Oxymercuration-Demercuration)
NaBHfour also makes an appearance in the oxymercuration reaction. Specifially, NaBHiv is used in the second step of the reaction, to break the C-Hg bond and plow it into a C-H bond.
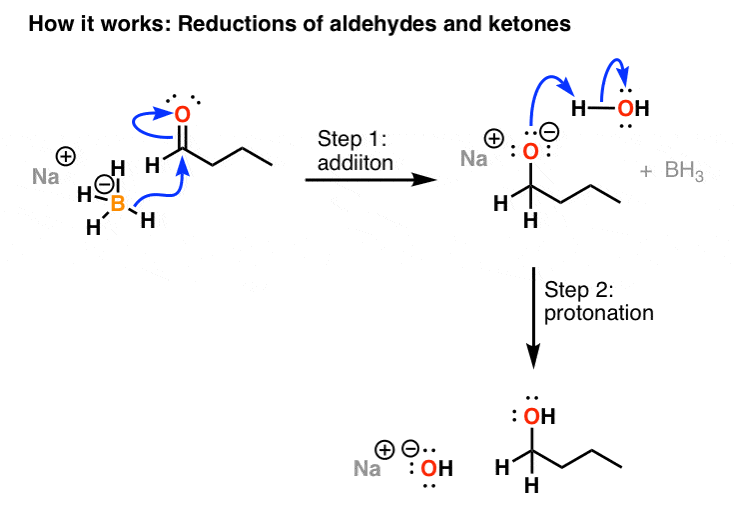
Mechanism For The Reduction Of Aldehydes And Ketones With NaBH4
How it works: The mechanism of the reaction of sodium borohydride with aldehydes and ketones gain in two steps. In the first step, H(–) detaches from the BHfour(–) and adds to the carbonyl carbon (an example of [1,2]-addition). This forms the C-H bond, and breaks the C-O bond, resulting in a new lone pair on the oxygen, which makes the oxygen negatively charged (FYI: we phone call these negatively charged oxygens alkoxides, as they are deprotonated alcohols). In the 2nd footstep, a proton from water (or an acid such every bit NH4Cl) is added to the alkoxide to brand the alcohol. This is performed at the end of the reaction, a stride referred to every bit the workup.
I suppose I should also mention that NaBH4 will reduce acyl halides to alcohols, just things are a petty lengthy here already.
Note: this machinery assumes a polar protic solvent. What if you use a slightly different solvent? You take a slightly different mechanism, run into notation below.
I also won't go into the detailed arrow pushing for NaBH4 in the oxymercuration reaction. Just the key indicate is that the carbon-mercury bail is broken, and a new carbon hydrogen bond is formed, and it is NaBHfour which performs this reaction. It works out like this:
P.S. You tin can read most the chemistry of NaBHiv and more than 80 other reagents in undergraduate organic chemistry in the "Organic Chemical science Reagent Guide", available hither as a downloadable PDF.
![]()
Notes
A notation nigh the mechanism.
The machinery drawn above works in a polar protic solvent similar methanol, which can protonate the alkoxide. What happens if you use a not-protic solvent like DMF? Well, since you don't have a proton source, you form a salt! It would look something like this:
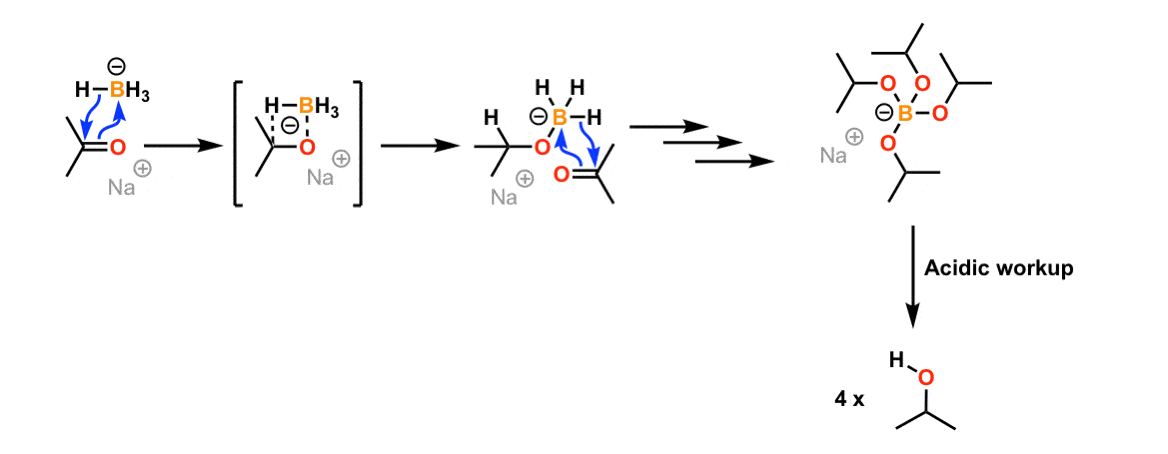
This article has more item:
Reduction of ketones by sodium borohydride in the absence of protic solvents. Inter versus intramolecular mechanism.
Kayser, Grand., Eliev, South., & Eisenstein, O.
Tetrahedron Messages,1983 24(ten), 1015–1018.
DOI: 10.1016/S0040-4039(00)81590-7
(Avant-garde) References and Further Reading:
- First example
Brown, H.C.; Wheeler, O.H.; Ichikawa, K.
Tetrahedron 1:214 (1957)
DOI: ten.1016/0040-4020(57)88041-7
Early paper by Nobel Laureate H. C. Brownish describing the reactivities of elementary aldehydes and ketones to reduction by NaBHfour, in which it is shown that aldehydes are more than reactive than ketones to nucleophilic reactions. - Mechanistic studies
Chocolate-brown, H. C.; Ichikawa, K.
Tetrahedron 1:221 (1957)
DOI: 10.1016/0040-4020(57)88042-nine
This paper and the above are both mechanistic studies on the reduction of carbonyls – this paper investigates the effect of ring size on the reduction of cyclic ketones (e.one thousand. reduction of cyclobutanone vs. cyclopentanone, cyclohexanone, etc.). - Reference To An Experimental Procedure
Antonio Bermejo Gómez, Nanna Ahlsten, Ana Eastward. Platero-Prats and Belén Martín-Matute
Org. Synth. 2014, 91, 185
DOI: 10.15227/orgsyn.091.0185
The first step in this procedure uses NaBH4 to reduce a cinnamyl ketone to the alcohol.
rowlettfetwerivid.blogspot.com
Source: https://www.masterorganicchemistry.com/2011/08/12/reagent-friday-sodium-borohydride-nabh4/
0 Response to "Benzoin Can Be Reduced With Reagents Such as Nabh4. What Stereochemical Products Are Possible"
Post a Comment